| Intended Use |
The Canine Adenovirus Type-I Virus Antigen Rapid Test Device is for the qualitative detection of Canine Adenovirus Type-I Virus from swab specimens.
The Canine Adenovirus Type-I Virus Antigen Rapid Test Device is a rapid chromatographic immunoassay for the qualitative detection of Canine Adenovirus Type-I Virus Antigen in nasopharyngeal secretions.
The Canine Adenovirus Type-I Virus Antigen Rapid Test Device is a rapid test to qualitatively detect the presence of CAV I in nasopharyngeal secretions or serum.
For professional in vitro diagnostic use only.
| What is the Canine Adenovirus 1 (CAV-1)? |
Canine Adenovirus 1 (CAV-1) was first isolated by Carbasso in 19541 from a case of acute hepatitis in the dog. This virus found to be identical to the virus isolated in 1947 by Rubarth2 from a dog showing acute liver lesions, and so CAV-1 was originally known as Infectious Canine Hepatitis (ICH) virus. Subsequently, CAV1 infection was shown to be common in young dogs worldwide, with 82% of British dogs displaying neutralising antibody titres by nine months of age3. It has also since been demonstrated that CAV1 has a role in diseases other than Infectious Canine Hepatitis, such as Canine Infectious Tracheobronchitis.
CAV-1 is a Mastadenovirus, member of the Adenoviridae family.
 | Pathology of the Canine adenovirus 1 |
Canine adenovirus 1 subclinical infection normally results in a moderate bronchointerstitial pneumonia, while immunocompromised dogs can develop a necrotizing bronchiolitis. Histologically, bronchointerstitial pneumonia is characterized by pulmonary oedema, proliferation of type II pneumocytes, and necrosis of the bronchiolar and alveolar epithelium.
Canine adenovirus 1 primarily damages the endothelium and hepatic cells in Infectious Canine Hepatitis. Hepatic injury can be seen as an enlarged liver with regions of necrosis and endothelial damage as extensive petechial hemorrhages. Adenoviral nuclear inclusion bodies may be identified in Kupffer and parenchymal cells, and centrolobular necrosis is seen in the liver under a microscope. Occulular pathology and glomerulonephritis are frequent findings.
| Principle |
Canine Adenovirus Type-I Virus Antigen Rapid Test Device is based on sandwich lateral flow immunochromatographic assay. The Canine Adenovirus Type-I Virus Antigen Rapid Test Device device has one testing window. Each testing window has an invisible T (test) zone and C (control) zone.
When the sample is applied into the sample hole on the Canine Adenovirus Type-I Virus Antigen Rapid Test Device, the liquid will laterally flow on the surface of the Canine Adenovirus Type-I Virus Antigen Rapid Test Device strip. If there is enough CAV-I antigen in the sample, a visible T band will appear in the corresponding testing window. The C band should always appear after a sample is applied, indicating a valid result.
By this means, the Canine Adenovirus Type-I Virus Antigen Rapid Test Device can accurately indicate the presence of CAV-I antigen in the sample.

| Test Procedure |
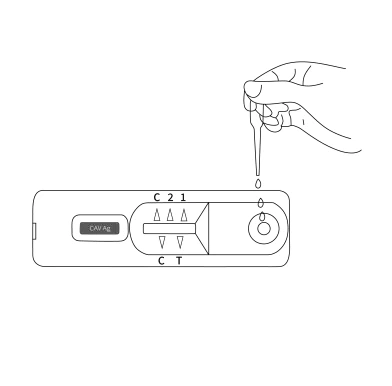
Place all specimens, Canine Adenovirus Type-I Virus Antigen Rapid Test Device, and allow them to room temperature prior to testing.
Remove the Canine Adenovirus Type-I Virus Antigen Rapid Test Device from the sealed pouch, and place it on a clean and plain place.
Collect dog’s nasopharyngeal secretions with the swab stick. Make the swab wet sufficiently. Insert the wet swab into the provided assay buffer tube. Agitate it to assure good sample extraction.
Within 5~10 minutes, interpret the test result. Do not read the result after 20 minutes.
| Interpretation of Result |
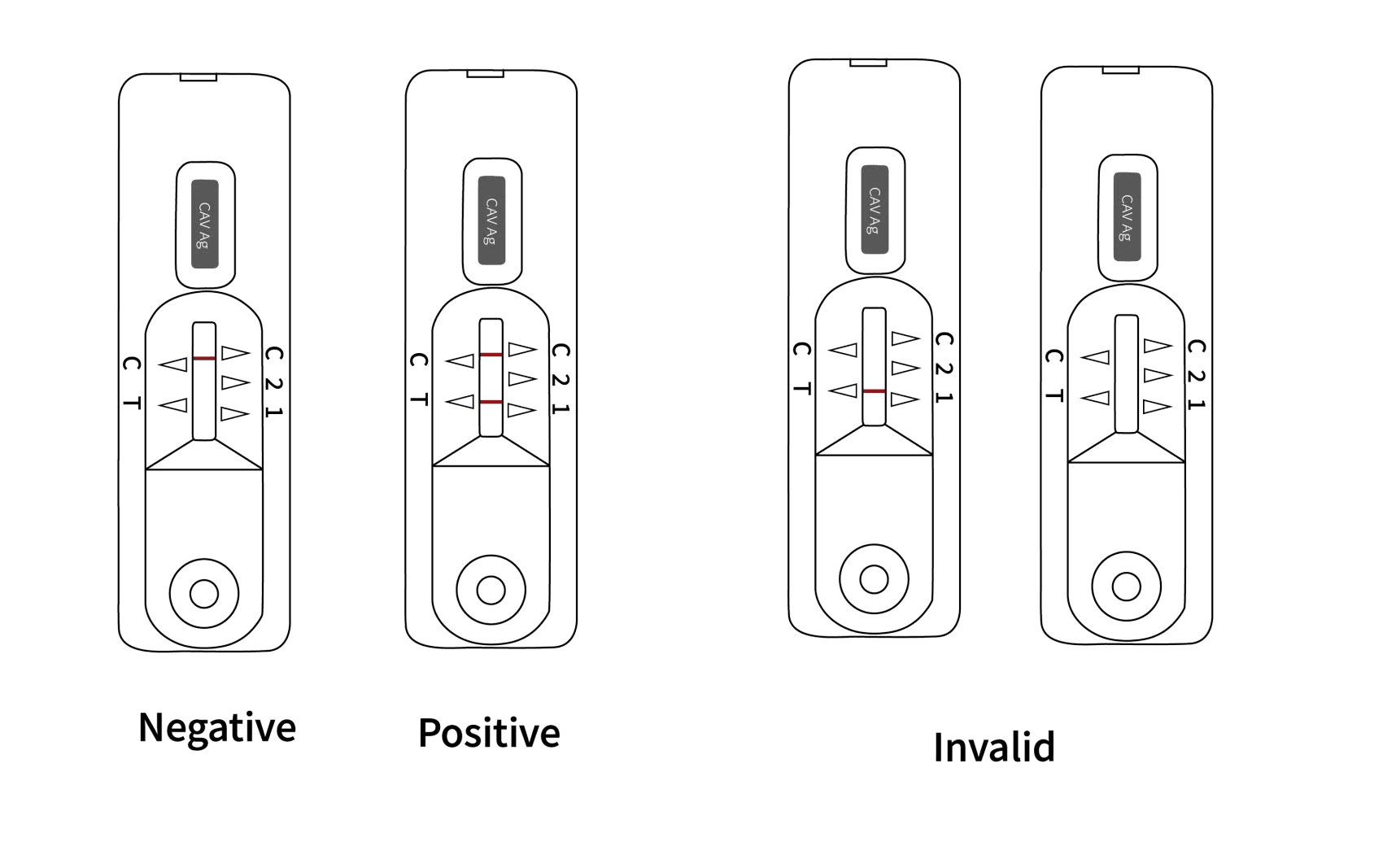
 | POSITIVE |
Two distinct colored lines appear on the Canine Adenovirus Type-I Virus Antigen Rapid Test Device. One line should be in the control region (C) and another line should be in the Canine Adenovirus Type-I Virus Antigen Rapid Test Device region (T).
NOTE: The intensity of the color in the Canine Adenovirus Type-I Virus Antigen Rapid Test Device line region (T) will vary depending on the concentration of the nasopharyngeal secretions or serum CAV I present in the specimen. Therefore, any shade of color in the test region (T) should be considered positive.
 | NEGATIVE |
One colored line appears in the control region (C). No apparent colored line appears in the test region (T).
 | INVALID |
Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques are the most likely reasons for control line failure. Review the procedure and repeat the test with a new Canine Adenovirus Type-I Virus Antigen Rapid Test Device.
| Performance Characteristics |
 | Sensitivity |
The Canine Adenovirus Type-I Virus Antigen Rapid Test Device has been tested with a positive panel and compared with a leading commercial CAV I rapid test using clinical specimens. The results show that the relative sensitivity of the Canine Adenovirus Type-I Virus Antigen Rapid Test Device is over 99.0%. Rapid Test Device has been tested with a positive panel and compared with a leading commercial CAV I rapid test using clinical specimens. The results show that the relative sensitivity of the Canine Adenovirus Type-I Virus Antigen Rapid Test Device is over 99.0%.
 | Specificity |
Antibodies used for the Canine Adenovirus Type-I Virus Antigen Rapid Test Device were developed against CAV I Antigen Rapid Test Device was also tested with laboratory strains of CPV and CCV. They all yielded negative results.
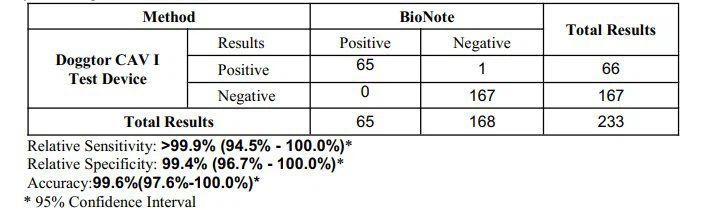
 | Precision of the Canine Adenovirus Type-I Virus Antigen Rapid Test Device |

Intra-Assay:
Within-run precision has been determined by using 10 replicates of 4 specimens: a negative, a low positive, a medium positive and a high positive. The negative, low positive, medium positive and high positive values were correctly identified >99% of the time.
Inter-Assay:
Between-run precision has been determined by 10 independent assays on the same 4 specimens: a negative, a low positive, a medium positive and a high positive. Three different lots have been tested using negative, low positive, medium positive and high positive specimens. The specimens were correctly identified >99% of the time.
Why choose us?
- If you need reliable and efficient Rapid Test Diagnostics products, look no further than our Chinese factory.
- In the absence of 'craftsman spirit" and "contract spirit' business environment, our company is committed to starting from a product to create a valuable product in the true sense.
- We are committed to providing our clients with excellent service and high-quality Rapid Test Diagnostics products.
- Based on deep thinking and far-sighted foresight of domestic and international market competition, our company relies on a good platform of product innovation, research and development and production technology to gather intelligence, build an elite team and implement lean management to meet the greater challenges of the market.
- Our manufacturing process is highly efficient, ensuring that we can deliver high-quality Rapid Test Diagnostics products quickly.
- It has been proved that the first step of employing talents is the first step of the enterprise, which is also a crucial step.
- Our team is dedicated to providing excellent customer service and support for all of our Rapid Test Diagnostics products.
- We always strictly implement the regulations of the national and local governments and environmental protection departments, and adhere to the concept of environmental protection.
- We are both a supplier and a manufacturer of Rapid Test Diagnostics products, and we offer competitive pricing.
- Persisting in 'High quality, Prompt Delivery, Competitive Price', we have established long-term cooperation with clients from both overseas and domestically and get new and old clients' high comments for Canine Adenovirus I Virus Antigen Rapid Test Device.
Hot Tags: canine adenovirus i virus antigen rapid test device, China canine adenovirus i virus antigen rapid test device manufacturers, suppliers